|
Putting these pieces together gives the name carbon tetrachloride for this compound. The second element, chlor ine, becomes chlor ide, and we attach the correct numerical prefix (“tetra-”) to indicate that the molecule contains four chlorine atoms. The name begins with the name of the first element-carbon. Ionic compounds contain ions and are held together by the attractive forces among the oppositely charged ions. 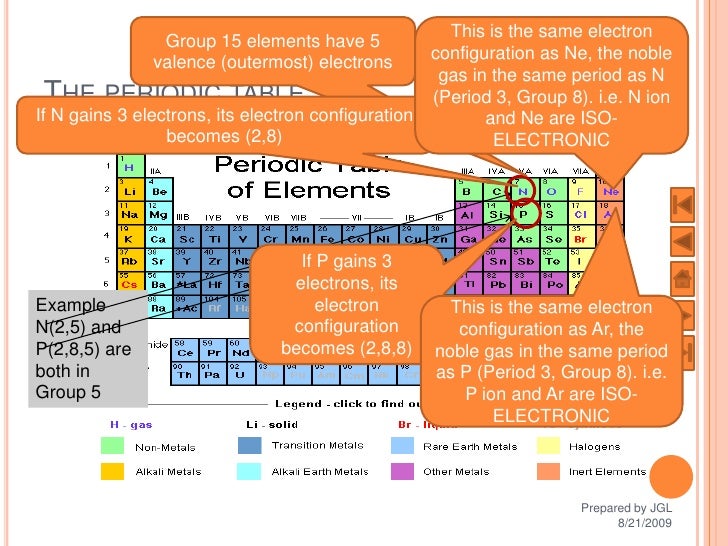
The resulting compound is called an ionic compound. Another classification scheme for chemical compounds is based on the types of bonds that the compound contains. Ionic bonding When metals react with non-metals, electrons are transferred from the metal atoms to the non-metal atoms, forming ions. 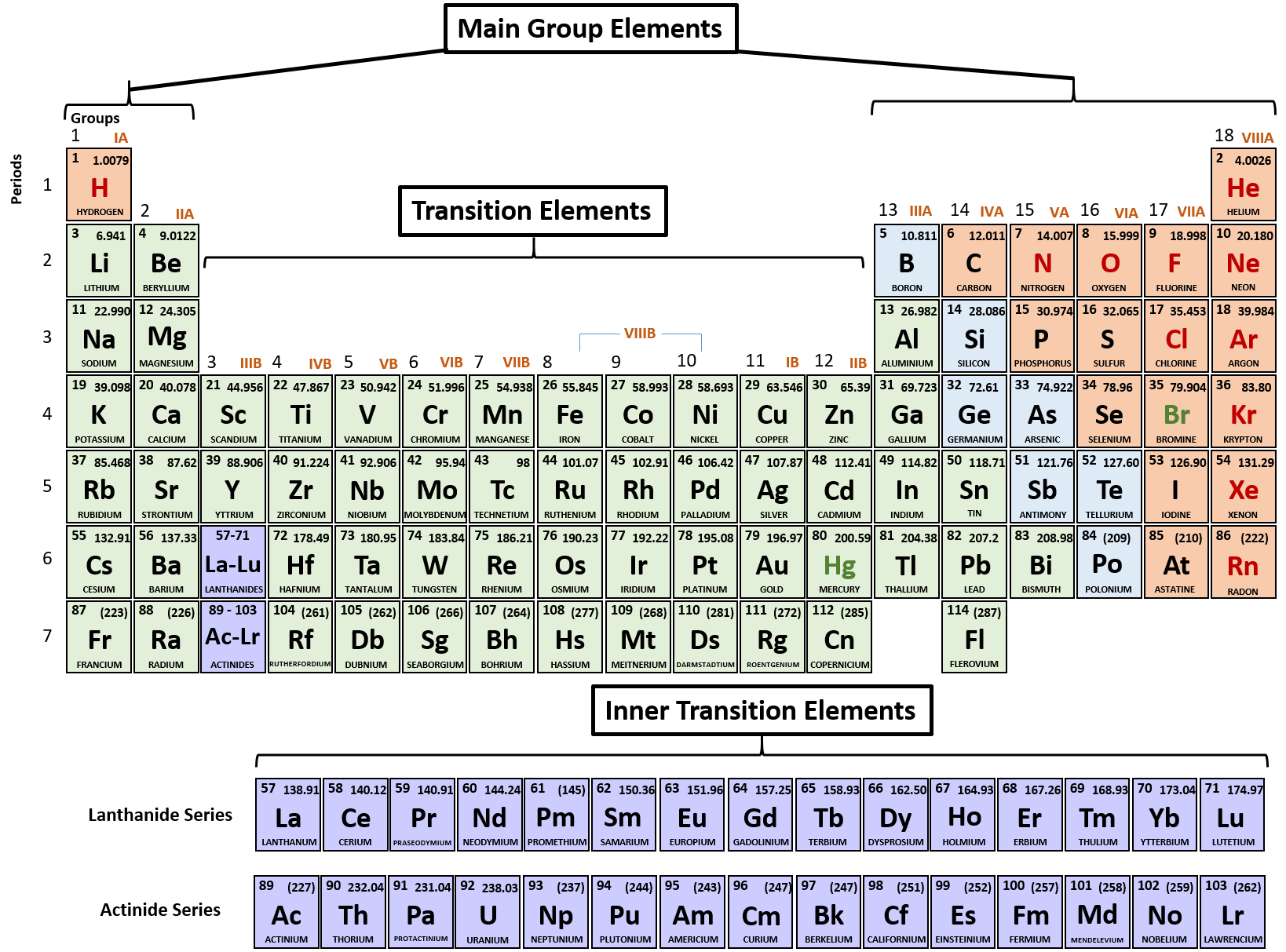
Let us practice by naming the compound whose molecular formula is CCl 4. As the name suggests, organometallic compounds are organic compounds bonded to metal atoms. *This prefix is not used for the first element’s name. The formula of the carbonate ion is CO 3 2. The carbonate ion (see figure below) consists of one carbon atom and three oxygen atoms and carries an overall charge of 2. 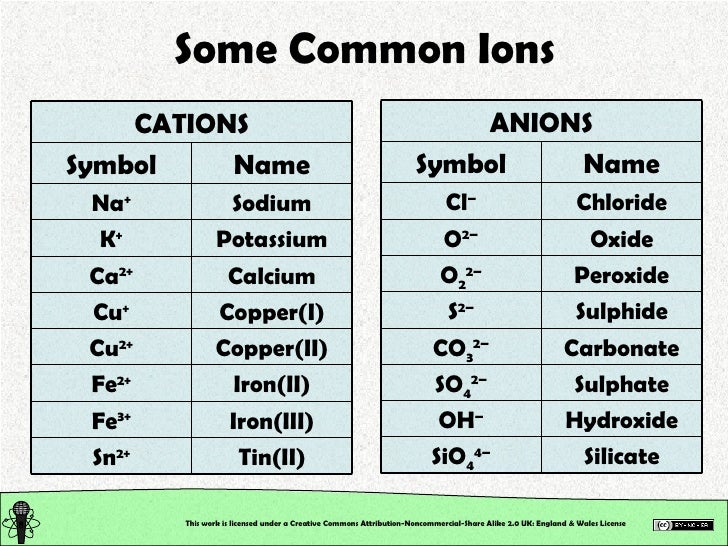
Diamond consists of carbon atoms that are covalently bonded in a network. Together, they comprise a single ion with a 1 charge and a formula of NH 4 . These all have ionic bonding in which metal atoms transfer valence electrons. \): Numerical Prefixes for Naming Binary Covalent Compounds Number of Atoms in Compound The ammonium ion (see figure below) consists of one nitrogen atom and four hydrogen atoms.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
